By Patricia Tolson
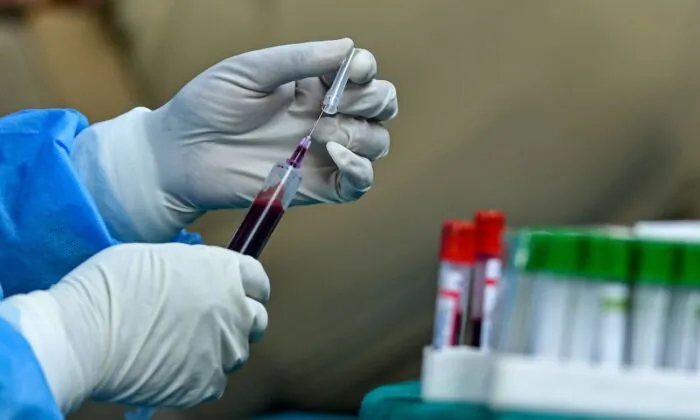
The demand for unvaccinated blood is on the rise, a blood products and services provider said.
Kirby Winn, public relations manager of ImpactLife, says that while the hospitals his company serves have not expressed any interest in receiving unvaccinated blood, he has noticed the demand for “pure blood” rising from the general population.
According to its website, ImpactLife provides blood products and services to more than 120 hospitals in Illinois, Iowa, Missouri, and Wisconsin.
“We have a lot of conversations with people who have questions and concerns about this,” Winn told The Epoch Times.
However, he acknowledged that those who ask questions about receiving blood from vaccinated donors “might not be satisfied with the answers.”
In a joint statement (pdf) issued on Jan. 26, the Association for the Advancement of Blood and Biotherapies, America’s Blood Centers, and the American Red Cross vouched for the safety of America’s blood supply, assuring the public that “vaccines do not pose a risk to patients receiving blood transfusions.”
As Winn explained, blood providers nationally “are confident that this is the right position” and they “continue to monitor blood transfusion-related incidents with the recipients of blood transfusions through a process called hemovigilance.”
“As far as the medical treatment goes, it’s not relevant if the donor is vaccinated or not vaccinated for COVID and we don’t track it,” Winn said. “When people come in to give blood we don’t ask the question, ‘Have you received a COVID vaccine or have you not received a COVID- vaccine.’”
Vaccinated Donors
The Jan. 26 joint statement also assured the public that because “there is no scientific evidence that demonstrates adverse outcomes” from blood transfusions from vaccinated donors, there is “no medical reason to distinguish or separate blood donations from individuals who have received a COVID-19 vaccination.”
However, the Red Cross states on its website that donors who received a COVID-19 vaccine will “need to provide the manufacturer name” when they come to donate.
“Upon vaccination, you should receive a card or printout indicating what COVID-19 vaccine was received, and we encourage you to bring that card with you to your next donation,” the Red Cross advises. “In most cases, there is no deferral time for individuals who received a COVID-19 vaccine as long as they are symptom-free and feeling well at the time of donation.”
While the FDA updated its information on Jan. 11, 2022, saying it “does not recommend using COVID-19 laboratory tests to screen routine blood donors,” it also said that “the blood establishment’s responsible physician must evaluate prospective donors and determine eligibility.”
The FDA further advised that donor candidates who were diagnosed with or tested positive for COVID-19 should “refrain from donating blood for at least 10 days after complete resolution of symptoms.” However, “individuals who are tested and found positive for SARS-CoV-2 antibodies” in their blood “can donate without a waiting period and without performing a diagnostic test.”
The coronavirus disease, commonly known as COVID-19, is caused by the SARS-CoV-2 virus and originated in Wuhan, China.
The COVID-19 pandemic had a devastating effect on the entire world. The protocols that were instituted to mitigate the deadly impact of COVID-19 caused additional damage.
Masking and social isolation measures divided America’s population into two camps. Those who embraced the mandates became intolerant of those who rejected them, and vice versa. According to data compiled by USA Facts, approximately 81 percent of Americans have received at least one dose of a COVID-19 vaccine as of May 10. About 70 percent are considered fully vaccinated. Those vaccines triggered a whole new era of segregation: the vaccinated versus the unvaccinated.
Blood Clots
The safety of COVID-19 vaccines became a highly-controversial issue after they were rolled out.
An analysis (pdf) regarding the demand for unvaccinated blood, issued Dec. 17, 2022, noted that “embalmers have reported finding unusual clots not only in deceased people” who’ve received COVID-19 vaccinations “but also in those who have had a blood transfusion.”
The analysis included comments from Steve Kirsch, executive director of the Vaccine Safety Research Foundation, who concluded that “the risk is not zero.”
A study led by the State University of New York at Buffalo (UB) and published on Feb. 1 in the Journal of Clinical and Translational Science confirmed that COVID-19 vaccines do pose a small risk of venous thromboembolism (VTE), or blood clots.
Dr. Peter L. Elkin, first author of the paper and a UB distinguished professor, called the risk “trivial,” noting that “about 1.4 cases per million” vaccinated patients were affected.
Mark Sherwood, a naturopathic doctor with the Functional Medicine Institute, suggests other factors are involved.
“I have talked to morticians who have been in the business for 25 and 30 years and they are indeed seeing an increase in clots in the bodies of both vaccinated and unvaccinated people,” Sherwood told The Epoch Times. “Further, they indicate to me that the question of vaccination or not is coming up more.”

Sherwood also said he “would be remiss” if he didn’t mention that the American diet is “creating massive inflammation on an organ system called the endothelium, which lends itself to more damage and more clot risk.”
He also noted the rise in cases of heart disease and vascular disease.
“That’s a very important point,” Sherwood insisted. “We need to look at all angles of this from both a medical and a journalistic standpoint.”
FDA Revises Major Donor Eligibility Rule
On May 11, the FDA finalized recommendations “assessing blood donor eligibility using a set of individual risk-based questions to reduce the risk of transfusion-transmitted HIV.”
“These final recommendations are consistent with the policy initially proposed in January,” the FDA said.
“This policy eliminates time-based deferrals and screening questions specific to men who have sex with men (MSM) and women who have sex with MSM,” the FDA advised.
Under the final guidance, all questions “will be the same for every donor, regardless of sexual orientation, sex or gender,” and blood-related establishments are advised that they “may now implement these recommendations by revising their donor history questionnaires and procedures.”
“All prospective donors who report having a new sexual partner, or more than one sexual partner in the past three months, and anal sex in the past three months, would be deferred to reduce the likelihood of donations by individuals with new or recent HIV infection who may be in the window period for detection of HIV by nucleic acid testing,” the FDA advised.
People taking medications to treat or prevent HIV infection will also be deferred.
While the FDA has issued final guidance, Winn said the changes have not yet been implemented.
“We have many changes to make to our donor screening system, donor records, and staff training,” Winn explained. “Even though the new guidance has been out for a couple of weeks, blood providers are not able to make these changes overnight. We are beginning the process, and welcome the change, but we have a lot of i’s to dot and a lot of t’s to cross.”
Winn anticipates the new eligibility criteria should be running by the end of the year.

High Risk of Transmission
By using individual risk-based questions, the FDA hopes to “reduce the risk of transfusion-transmitted HIV.” However, the Centers for Disease Control and Prevention (CDC) says blood transfusions pose the highest rate of HIV transmission per exposure.
For every 10,000 exposures through a blood transfusion, 9,250 recipients of that blood will contract HIV.
In a statement (pdf) issued on May 17, the Association for the Advancement of Blood and Biotherapies (AABB) joined the American Red Cross to “applaud the important, science-driven step forward in blood donation criteria that will base eligibility on an individual donor assessment using the same set of questions for all donors, regardless of sexual orientation.”
“The blood community is proud to mark this progress towards a more inclusive blood donation process and is committed to working with the FDA and other stakeholders to ensure that deferral changes continue to be examined moving forward.”
Sherwood said he is “very concerned” about the FDA’s decision to revise the screening requirements for assessing the eligibility of donors.
He believes the FDA’s new guidance is more of “a political stunt” than an effort to protect the public.
“When we start compromising principles for political correctness, we’re losing our direction,” Sherwood suggested. “This is a natural next step in the regressive progression.”
Sherwood warned that the new guidance could have devastating consequences.
“What if one HIV-infected donation slips through?” he asked. “While some will argue HIV is very treatable today, why would you be willing to pass it on to someone just because it’s easier to treat?”