By Joseph Mercola
Iron accumulation in the brain is emerging as a key player in neurodegeneration that may lead to Alzheimer’s disease.
STORY AT-A-GLANCE
- An imbalance of iron (Fe) in the body can lead to ferroptosis, a programmed cell death pathway known to play a role in neurodegenerative diseases like Alzheimer’s.
- Alzheimer’s patients typically have elevated iron levels in the brain.
- In areas of the brain where amyloid beta plaques tend to accumulate, an increase in iron redox was revealed, suggesting that iron located in these areas became “more reactive in the presence of oxygen.”
- Iron accumulations in the brain tend to concentrate in areas most affected by Alzheimer’s, namely the frontal cortex and hippocampus.
- The APOE4 gene, which is considered to be the strongest risk factor for Alzheimer’s disease, is also known to elevate iron levels in the brain.
By 2050, an estimated 13 million Americans will have Alzheimer’s disease.1 But despite its prevalence, there’s no known cure, and its causes are hotly debated. While it’s often said that an accumulation of amyloid beta plaques underlies Alzheimer’s, there appears to be more to the story.
It’s been suggested the plaques may be a symptom of Alzheimer’s rather than its cause—and may even have a protective role.2 Iron in the brain may also be involved, according to a team of researchers from The University of Texas (UT) at Austin and the University of Illinois at Urbana-Champaign.3
Iron’s Link to Alzheimer’s
An imbalance of iron (Fe) in the body can lead to ferroptosis, a programmed cell death pathway known to play a role in neurodegenerative diseases like Alzheimer’s.4 Previous research also suggests Alzheimer’s patients typically have elevated iron levels in the brain, but at the time there was no way to measure two different forms of iron (Fe2+ and Fe3+).
The featured study, published in Science Advances,5 changed that, as the team developed DNA-based fluorescent sensors capable of detecting Fe2+ and Fe3+ in animal studies. The sensors glow different colors for each type of iron, allowing researchers to see their quantity and how they’re distributed in the brain.6 Study author Yuting Wu with UT Austin explains:7
“The best part about our sensor is that we can now visualize the changes of Fe2+ and Fe3+ and their ratios in each location. We can change one parameter at a time to see if it changes the plaques or the oxidative states of iron.”
The tests revealed “a decreased Fe3+/Fe2+ ratio during ferroptosis and an increased Fe3+/Fe2+ ratio in Alzheimer’s disease.” Further, the team notes, “The elevated Fe3+/Fe2+ ratio was mainly observed in amyloid plaque regions, suggesting a correlation between amyloid plaques and the accumulation of Fe3+ and/or conversion of Fe2+ to Fe3+.”8
In areas of the brain where amyloid beta plaques tend to accumulate, an increase in iron redox was revealed, suggesting that iron located in these areas became “more reactive in the presence of oxygen.”9 The team plans to further investigate if iron and redox changes cause cell death in Alzheimer’s, and whether triggering changes in the Fe3+ to Fe2+ ratio could be protective. The team concluded:10
“Our data suggest that not only total iron but also iron redox cycling is involved in the progression of AD [Alzheimer’s disease]. Combining these data with our observation that both Fe2+ and Fe3+ levels increased around Aβ plaque regions and suggests a potential role of Aβ plaques in accumulating Fe3+ over Fe2+ from surrounding cells and/or proteins in AD mouse brains …
“However, it is unknown whether the dysregulated iron is involved in amyloid plaque formation, or this is a secondary effect of amyloid plaque formation …”
More Clues to the Iron-Alzheimer’s Connection
While iron plays an important role in brain activities such as neurotransmitter synthesis, myelination, and mitochondrial function, it can also be a source of oxidative stress. Iron accumulation in the brain, which may occur with aging, may be a contributing factor to neurodegeneration. It’s unknown why iron accumulates in the brain with age, but it may be linked to inflammation.11
Further, studies show that iron accumulations in the brain tend to concentrate in areas most affected by Alzheimer’s, namely the frontal cortex and hippocampus. Magnetic resonance imaging tests have also revealed elevated iron in brains affected by Alzheimer’s. Cerebrospinal fluid ferritin may also be a proxy for iron accumulation in the brain, with one study finding it to be predictive of cognitive decline over the next seven years.12
Ferritin is a protein that’s the carrier molecule of iron. Plasma ferritin also tends to be elevated in patients with Alzheimer’s disease, while the APOE4 gene, which is considered to be the strongest risk factor for Alzheimer’s disease,13 is also known to elevate iron levels in the brain.14
In fact, elevated levels of iron in your brain may actually be the mechanism that makes APOE4 a major genetic risk factor for the disease.15 Writing in the Journal of Biological Chemistry, researchers explained that iron could contribute to Alzheimer’s in multiple ways, including:16
- Driving the formation of plaques and tangles.
- Promoting amyloid beta aggregation.
- Triggering neuronal toxicity.
“Taken together,” the team explains, “these findings build a case for how iron, either building up in the tissue, bound to the amyloid or tangle proteinopathy, inducing the proteinopathy, or in tandem with the proteinopathy, might contribute to AD pathophysiology. Targeting iron, therefore, might be a therapeutic strategy for AD.”17
Why Clearing Out Excess Iron Is Important for Your Brain
According to research published in 2018, buildup of iron has a type of “rusting effect” in your brain and is common in most Alzheimer’s patients. As noted by the authors:18
“In the presence of the pathological hallmarks of [Alzheimer’s disease], iron is accumulated within and around the amyloid-beta plaques and neurofibrillary tangles, mostly as ferrihydrite inside ferritin, hemosiderin, and magnetite.
“The co-localization of iron with amyloid-beta has been proposed to constitute a major source of toxicity. Indeed, in vitro, amyloid-beta has been shown to convert ferric iron to ferrous iron, which can act as a catalyst for the Fenton reaction to generate toxic free radicals, which in turn result in oxidative stress.”
Iron-chelating strategies have shown some promise in improving Alzheimer’s symptoms,19 but research remains limited, largely because to date the focus has been placed on strategies to reduce amyloid beta, with little to show for it. But, researchers wrote in the International Journal of Molecular Sciences, understanding the “metabolic balance mechanism of iron in the brain” is critical for Alzheimer’s treatment:20
“In AD, oxidative stress caused by brain iron accumulation promotes the deposition of amyloid protein and the hyperphosphorylation of tau, which causes damage to neurons, resulting in declines in motor, cognitive, and memory functions …
“Although using iron-chelating strategies has achieved some positive results for improving the symptoms of AD, there is still much research needed in order to translate the research into practice for the clinical treatment of AD.
“Nevertheless, there have been few studies on iron-reducing strategies in AD patients through genetic methods, and excessive emphasis has been put on the amyloid-reducing strategies, which have been disappointing thus far.
“Given that, more and more iron-chelating compounds have potential disease-improving effects, as well as the availability of biomarkers of iron load in MRI and cerebrospinal fluid, there is considerable room for exploring this type of treatment to avoid its side effects as far as possible.”
Iron Dysregulation Harms Your Health
Aside from its effects on your brain, iron buildup can be harmful elsewhere in your body as well. Your body has a limited capacity to excrete iron, so it can easily build up in organs like your liver, heart, and pancreas. This is dangerous because iron is a potent oxidizer that can damage your tissues and contribute to a variety of health problems, including cancer.
Elevated ferritin is associated with a 2.9 times higher risk of death from cancer21 and blood donors have been shown to have a lower likelihood of developing certain cancers than nondonors.22
Elevated ferritin has also been linked to dysfunctional glucose metabolism, raising the risk of diabetes nearly fivefold in men and fourfold in women.23,24 High ferritin also increases your risk of metabolic syndrome,25 a condition associated with an increased risk of high blood pressure, liver disease, heart disease, and insulin resistance.
How to Check Your Iron Levels
You can have your iron levels checked using a simple blood test called a serum ferritin test. I believe this is one of the most important tests that everyone should have done on a regular basis as part of a preventive, proactive health screen. If your ferritin levels are low, it means your iron levels are also low.
The healthy range of serum ferritin lies between 20 and 80 nanograms per milliliter (ng/ml). Below 20 ng/ml is a strong indicator that you are iron deficient, and above 80 ng/ml suggests you have an iron surplus. An ideal range is between 40 and 60 ng/ml.
Aside from a serum ferritin test, a gamma-glutamyl transpeptidase (GGT) test can also be used as a screening marker for excess free iron and is a great indicator of your risk for sudden cardiac death, insulin resistance, and cardiometabolic disease.26
In recent years, scientists have discovered GGT is highly interactive with iron. Low GGT tends to be protective against higher ferritin, so if your GGT is low, you’re largely protected even if your ferritin is a bit higher than ideal. When both your serum ferritin and GGT are high, you are at significantly increased risk of chronic health problems and early death.27
Where does iron overload come from? Aside from genetics, which can contribute to hereditary hemochromatosis, or iron overload, virtually all adult men and postmenopausal women are also at risk for iron overload since they do not lose blood on a regular basis. Blood loss is the primary way to lower excess iron.
Eating processed foods fortified with iron, taking iron-containing supplements, or cooking in iron pots and pans can also increase your risk of iron overload. Drinking well water that’s high in iron is also a risk, so make sure you have some type of iron precipitator and/or a reverse osmosis water filter. Regularly consuming alcohol is another risk factor, as this increases the absorption of iron from your diet.
A Simple Way to Reduce Iron Overload
The good news is that lowering your iron is easy. All you need to do is donate blood two to four times a year. You can also remove blood in smaller amounts once a month according to the schedule below. If you have congestive heart failure or severe COPD [Chronic Obstructive Pulmonary Disease], you should discuss this with your doctor, but otherwise, this is a fairly appropriate recommendation for most.
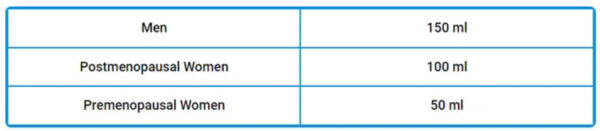
If, for some reason, a blood donor center is unable to accept your blood for donation, you can obtain a prescription for therapeutic phlebotomy. Lowering iron is one side of the equation. Iron and copper are highly interdependent and need to be considered together. Iron overload along with copper deficiency is a dangerous combination. Most people are deficient in copper and actually need more in order for their iron metabolism to function properly.
Depending on your copper levels, you may need to take up to 3 milligrams (mg) to 4 mg of copper bisglycinate per day or eat copper-rich foods, such as bee pollen, grass-fed beef liver, and acerola cherry. Acerola cherry is very high in vitamin C, which contains copper-rich tyrosinase enzyme.
Retinol, which makes copper bioavailable, is also important. It’s found in beef liver and beef organs, so if you eat that, you may not need any kind of supplement. Absent that, cod liver oil is a recognized source of real retinol.
Originally published May 17, 2023, on Mercola.com
◇ References:
- 1 Alzheimer’s Association, Facts and Figures, Quick Facts
- 2 J Alzheimers Dis. 2009 Oct; 18(2): 447–452
- 3, 6, 7, 9 EurekAlert April 19, 2023
- 4, 5 Science Advances April 19, 2023
- 8 Science Advances April 19, 2023, Abstract
- 10 Science Advances April 19, 2023, Discussion
- 11, 12, 14, 16, 17 J Biol Chem. 2021 Jan-Jun; 296: 100105., Iron
- 13 NIH, National Institute on Aging March 16, 2021
- 15 Nature Communications May 19, 2015
- 18 Scientific Reports 2018; 8: 6898, Introduction
- 19 Neurotherapeutics. 2021 Jan; 18(1): 252–264
- 20 Int J Mol Sci. 2021 Nov; 22(22): 12442
- 21 J Natl Cancer Inst. 1986 Apr;76(4):605-10
- 22 J Natl Cancer Inst. 2008 Apr 16;100(8):572-9
- 23 Diabetes Care 1997 Mar;20(3):426-8
- 24 Diabetes Care 1999 Dec;22(12):1978-83
- 25 Diabetes Care 2004 Oct;27(10):2422-8
- 26 European Journal of Preventive Cardiology 2014 Dec;21(12):1541-8
- 27 Journal of Insurance Medicine 2012;43(3):162-8
Views expressed in this article are the opinions of the author and do not necessarily reflect our views.